Q4 Savings on Clinically Characterized Samples — Reliable, Ready-to-Ship, and Designed to Support Your Research
Learn MoreApril 15, 2026
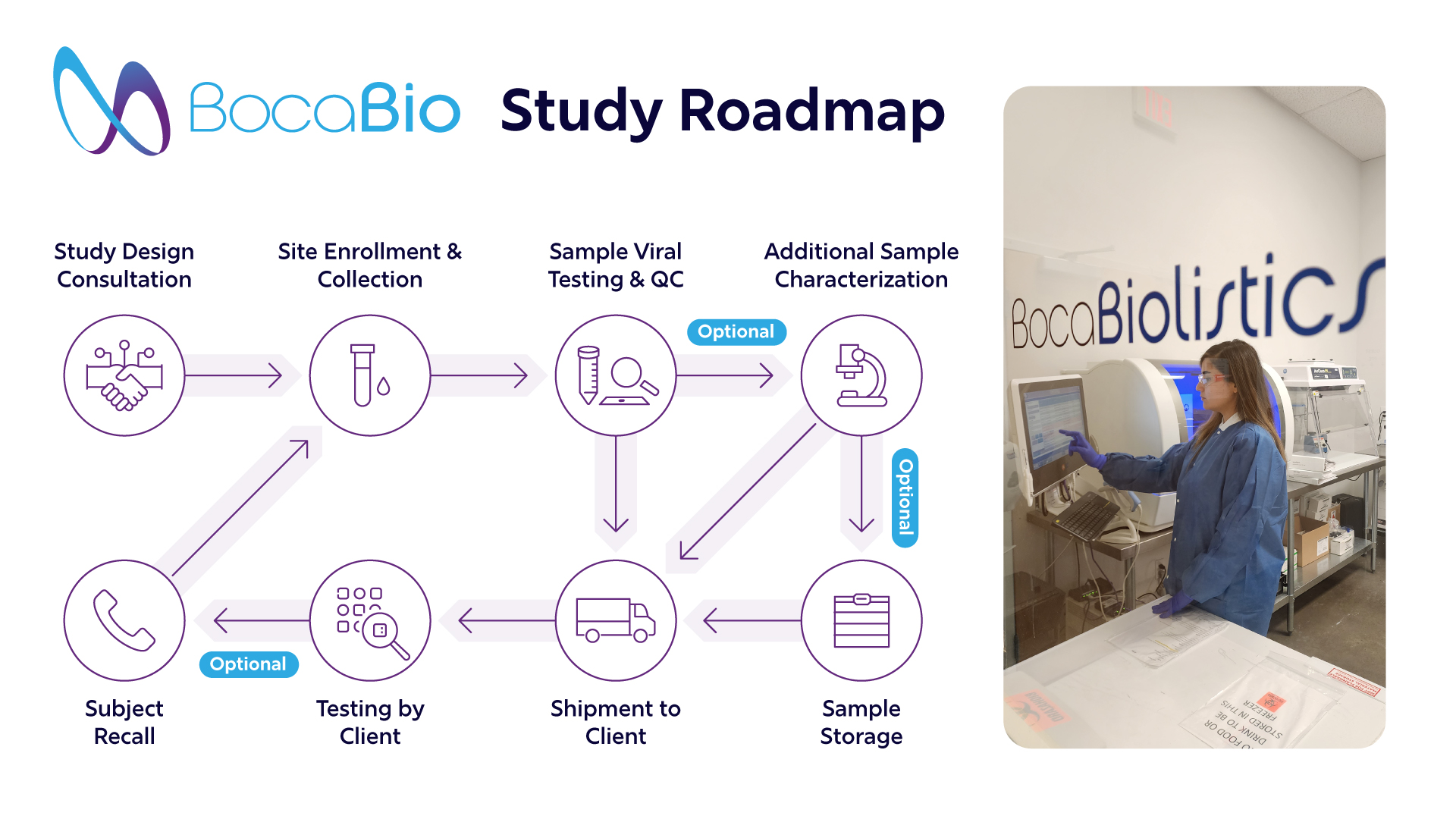
Capturing immune response data within the first 1–6 months post-immunization is essential, but recruiting qualified participants quickly can be costly and complex.
Boca Bio delivers streamlined, end-to-end serum procurement:
-300 serum samples collected in <4 months
-3 U.S. clinical sites
-Targeted enrollment of recently vaccinated adults
-Comprehensive metadata (demographics, vaccine status, medical history)
-CLIA- and CAP-certified biorepository storage
All samples undergo rigorous testing and are collected under strict quality and compliance standards.
-Rapid enrollment enabled study expansion from 200 → 300 samples
-Ongoing longitudinal follow-up with subject recall
-High-quality datasets to support biomarker analysis and vaccine benchmarking
-Fast turnaround for time-sensitive studies
-Scalable recruitment and collection
-21 CFR Part 11-compliant data systems
-HIPAA-compliant and ISO 13485:2016 certified
Bullet Point List Lorem ipsum dolor
Lorem ipsum dolor sit amet
Fusce tristique lorem non justo malesuada, et ullamcorper risus volutpat
Cras fringilla urna vel metus lobortis, eget egestas nulla efficias fringilla urna vel mngilla urna vel metus lobortis, eget egestas nulla efficias fringilla urna vel metus lobortie
Fusce tristique lorem non justo malesuada, et ullamcorper risus volutpat